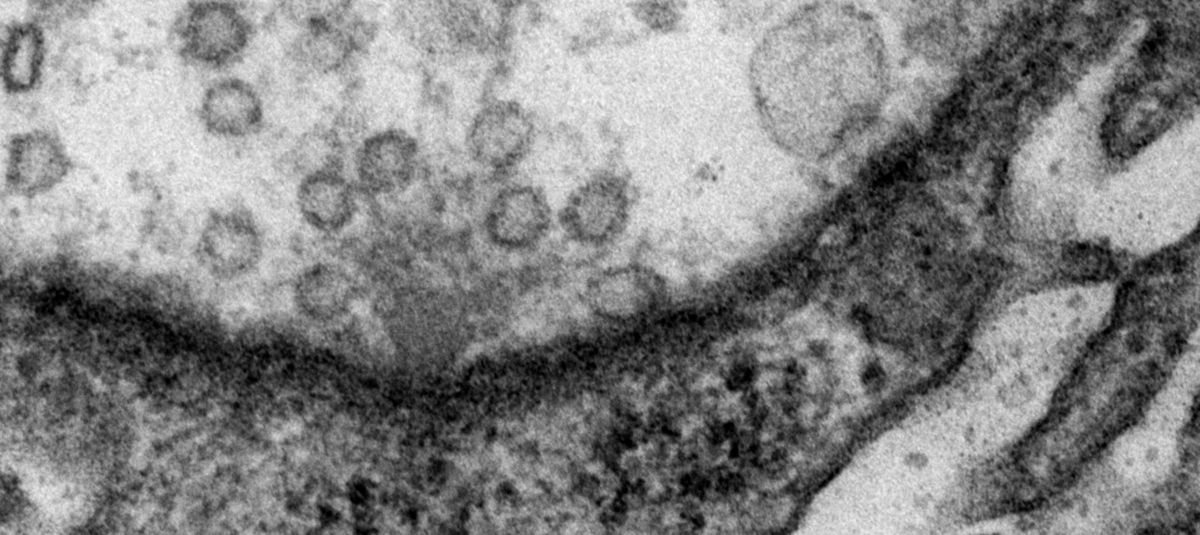
We study molecular mechanisms of neuronal communication. Our current research projects focus on a highly specialized and fascinating sub-cellular compartment: THE PRESYNAPTIC ACTIVE ZONE.
Como siempre, el genio de la vida, al construir sus obras portentosas, brilla en lo pequeño mucho mas aún que en lo grande.
Santiago Ramón y Cajal
Plasticity
Synapses are specialised intercellular contact sites, which serve as the communication link between neurons and their partner cells. At chemical synapses, calcium-ion influx triggers the fusion of transmitter-laden vesicles with the presynaptic membrane at a specific sub-cellular region termed the active zone. Transmitter substances released by this process then diffuse across the synaptic cleft and are sensed by postsynaptic receptors to convey signal transduction. A hallmark of synaptic transmission is its plasticity, which enables synapses to regulate complex brain processes by filtering, modifying, or integrating information. The details of active zone physiology and how its modulation contributes to synaptic plasticity are, however, far from understood.
By combining genetics with high resolution opto- and electrophysiological methods in Drosophila melanogaster (Dawydow et al., 2014; Ehmann et al., 2014), our research programme investigates how active zone physiology is modified by activity-induced plasticity in the behaving animal. To this end, molecular dynamics are followed at single synapse resolution and neurotransmission is both evoked and measured in an intact, alive, and genetically most amenable organism.
Our research projects follow a common strategy to link multiple levels of biological organisation. First, the experimentally accessible larval neuromuscular junction (NMJ) is employed to identify basic molecular underpinnings of active zone function and then the obtained results are transferred to the adult central nervous system (CNS). This approach enables us to study molecular mechanisms of active zone plasticity in the context of neuronal information coding and memory formation (Ehmann et al., 2017; Rozenfeld et al., 2023).
Pathology
Adhesion-type G protein-coupled receptors (aGPCRs) form a large class of seven- transmembrane spanning receptors involved in a number of critical developmental, immunological and neuronal processes (aGPCR Consortium). In collaboration with Tobias Langenhan, and supported by the former DFG-Research Group FOR 2149, we discovered that Latrophilin/CIRL, a prototype member of this receptor class, operates as a metabotropic mechanosensor in Drosophila (Scholz et al., 2015; Scholz et al., 2017).
Based on these findings, we have now extended our work to nociceptors (Dannhäuser et al., 2020). Together with our clinical colleague, Heike Rittner, we are pursuing a translational approach to investigate basic mechanisms of nociception and to explore the receptor's therapeutic potential. As part of the Clinical Research Unit KFO 5001, our research programme aims to identify further target molecules for nociceptor tuning and to examine the role of active zone components in neuropathy.
Evolution
In the frame of the DFG Priority Program "Evolutionary Optimization of Neural Processing" SPP 2205 our ongoing investigations have uncovered that different olfactory sensory neurons possess subtype-specific molecular organizations of the active zone. This finding is in line with recent work highlighting that active zones display considerable heterogeneities at functional and ultrastructural levels, not only between species and different cell types, but also between individual release sites of the same neuron.
It is thus an important research goal to identify evolutionarily conserved core features of the active zone and to distinguish these from specializations, which occur in adaptation to particular physiological demands. We are therefore exploring active zone properties in an evolutionary context and are extending our studies to early-branching animals.